
Results of interviews with high school chemistry students (N=14) during equation-solving sessions indicate that those who were able to construct diagrams consistent with notation of their balanced equation possessed good concepts of subscript and the balancing rule. Student Understanding of Chemical Equation Balancing. Presented is a method to teach this skill to high school chemistry students. Answers to a test on this topic were analyzed to determine the level of understanding and processes used by the students. (Contains 17 references.) (Author/YDS)ĭescribes a study of students' ability to balance equations. 
Surveys 13 methods, 6 based on matrix, 2 interactive programs, 1 stand-alone system, 1 developed in algorithm in Basic, 1 based on design engineering, 1 written in HyperCard, and 1 prepared for the World Wide Web. (Author/RT)Ĭomputer Applications in Balancing Chemical Equations.ĭiscusses computer-based approaches to balancing chemical equations. Additional specifications of courseware and availability information are included. Software consists of a compiled file of 46K for use under MS-DOS 2.0 or later on IBM PC or compatible computers. (TW)īALANCER: A Computer Program for Balancing Chemical Equations.ĭescribes the theory and operation of a computer program which was written to balance chemical equations. Provides examples and delineates the steps to follow for each type of equation balancing. Outlines a method which is based on changes in the oxidation number that can be applied to both molecular reactions and ionic reactions. Insights: A New Method to Balance Chemical Equations.ĭescribes a method designed to balance oxidation-reduction chemical equations. 
FORTRAN programs using this matrix method to chemical equation balancing are available from the author. Reviews mathematical techniques for solving systems of homogeneous linear equations and demonstrates that the algebraic method of balancing chemical equations is a matter of solving a system of homogeneous linear equations. You may also download just EBAS help file in Windows chm format.ERIC Educational Resources Information Center This is only a fraction of program features, feel free to browse the site to see more, or to download the free 30-day trial to try EBAS by yourself. Alternate interface can be used whenever there are too many reagents and the standard view doesn't fit on the screen. While there is no support for oxidation numbers or half reactions, in most cases to balance redox reaction it is enough to balance charge in addition to mass balance, thus most redox reaction will be balanced.įor calculation of amounts of substance present in solution aor in gaseous form, concentration and ideal gas calculators can be used.Īlternate user interface and reaction summary printout. If there are too many reagents and they don't fit the screen, you can use alternate user interface, shown on the third screen shot below.Īs you see program works with charged species as well as neutral molecules. 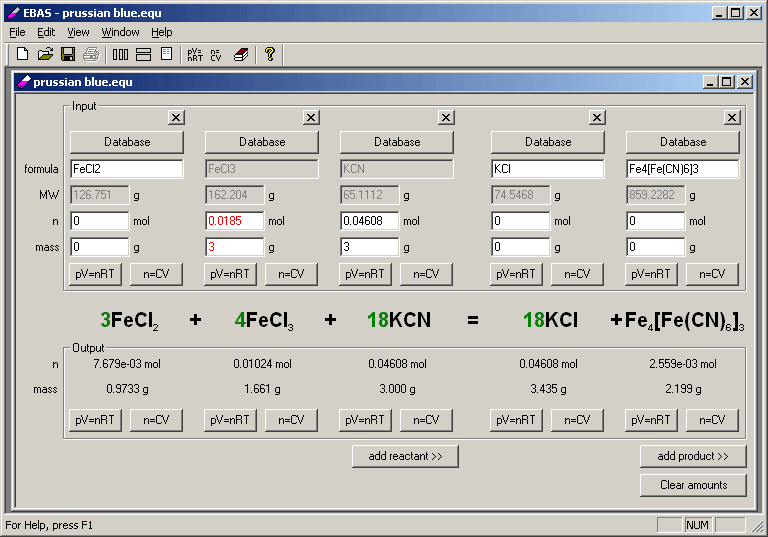
Every column contains information about one reagent. Balanced chemical equation is displayed in the middle. Upper part of the window (input frame) is where you enter known information, bottom part (output frame) is where you read calculation results. Size of the window (and number of reagents) can be changed using add reactant and add product buttons (or small crossed buttons above formula edit fields). This is the main program dialog - so called reaction view. Once you are done with the screen shots, select balancing equations to learn how to use dialog windows shown here. Equation Balancing and Stoichiometry calculator - scree nshots
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
